Research Overview
NAD+ Research Review: Cellular Energy, Mitochondrial Function, and Longevity Signaling
A detailed review of NAD+ research, redox metabolism, mitochondrial bioenergetics, sirtuin signaling, DNA repair biology, and growing interest in age-related cellular NAD+ decline.
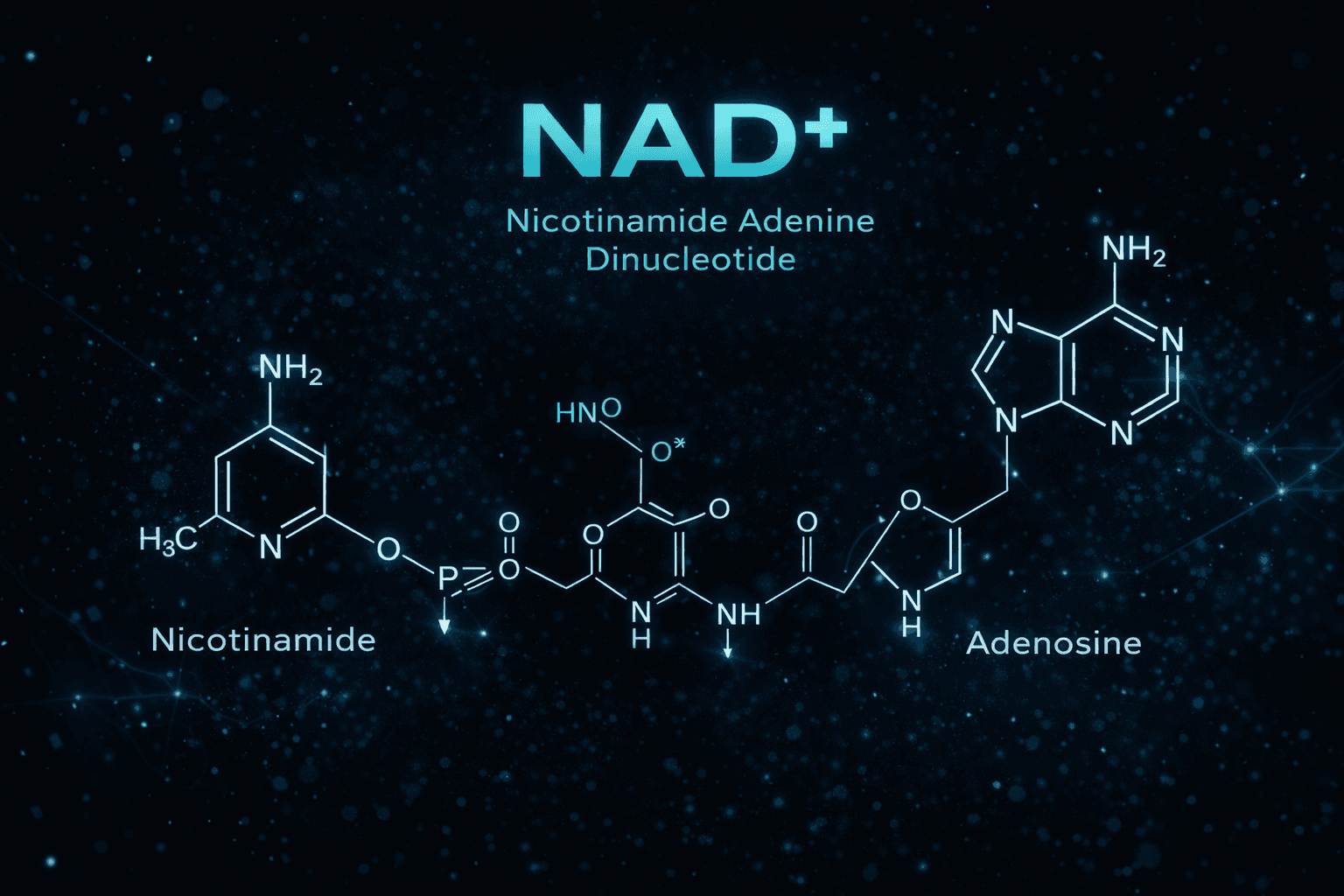
Abstract
Nicotinamide adenine dinucleotide (NAD+) is one of the most central metabolic cofactors in biology and is essential for cellular energy production, redox balance, and biochemical signaling. It functions as both an electron carrier and a substrate for major regulatory enzymes involved in DNA repair, stress adaptation, mitochondrial maintenance, and metabolic homeostasis.
Interest in NAD+ research has expanded significantly because its intracellular availability appears to influence mitochondrial performance, genomic stability, inflammatory signaling, and longevity-associated pathways. Research increasingly examines how declining NAD+ pools may contribute to metabolic dysfunction and how restoring NAD+ availability may support healthier cellular function in experimental models.
Introduction
NAD+ is present in every living cell and occupies a unique position at the intersection of metabolism and signaling. In its classical role, it participates in oxidation-reduction reactions that allow nutrients to be converted into usable energy. In its broader signaling role, it serves as a required substrate for enzymes that regulate cellular stress responses, chromatin dynamics, and DNA maintenance.
This dual function makes NAD+ unusually important in research focused on mitochondrial biology, aging, exercise adaptation, neurobiology, and metabolic resilience. Rather than being merely a background cofactor, NAD+ is increasingly understood as a dynamic molecule whose availability can shape large-scale physiological outcomes.
Because of that central role, NAD+ has become a major subject of investigation in studies examining mitochondrial output, redox balance, sirtuin activation, and cellular repair systems under both healthy and stressed conditions.
Molecular Properties
Structured molecular data helps place NAD+ within the broader category of metabolic and longevity-related compounds while making its biological role easier to understand in a research setting.
Compound Name
Nicotinamide adenine dinucleotide
Molecular Formula
C21H27N7O14P2
Molecular Weight
663.43 g/mol
Research Class
Cellular coenzyme / redox mediator
Molecular Structure and Functional Design
NAD+ is composed of two nucleotides joined by their phosphate groups: one containing adenine and one containing nicotinamide. The nicotinamide ring acts as the reactive redox center, allowing NAD+ to accept and donate electrons during metabolic reactions.
Research Focus
Redox Metabolism / Mitochondrial Function / Longevity Signaling
The significance of NAD+ lies not in receptor agonism but in its role as a universal metabolic currency that links nutrient oxidation, ATP generation, DNA repair processes, and cellular adaptation systems. Its chemistry allows it to function as both a metabolic intermediate and a signaling substrate, which is why it remains one of the most important molecules in cell biology.
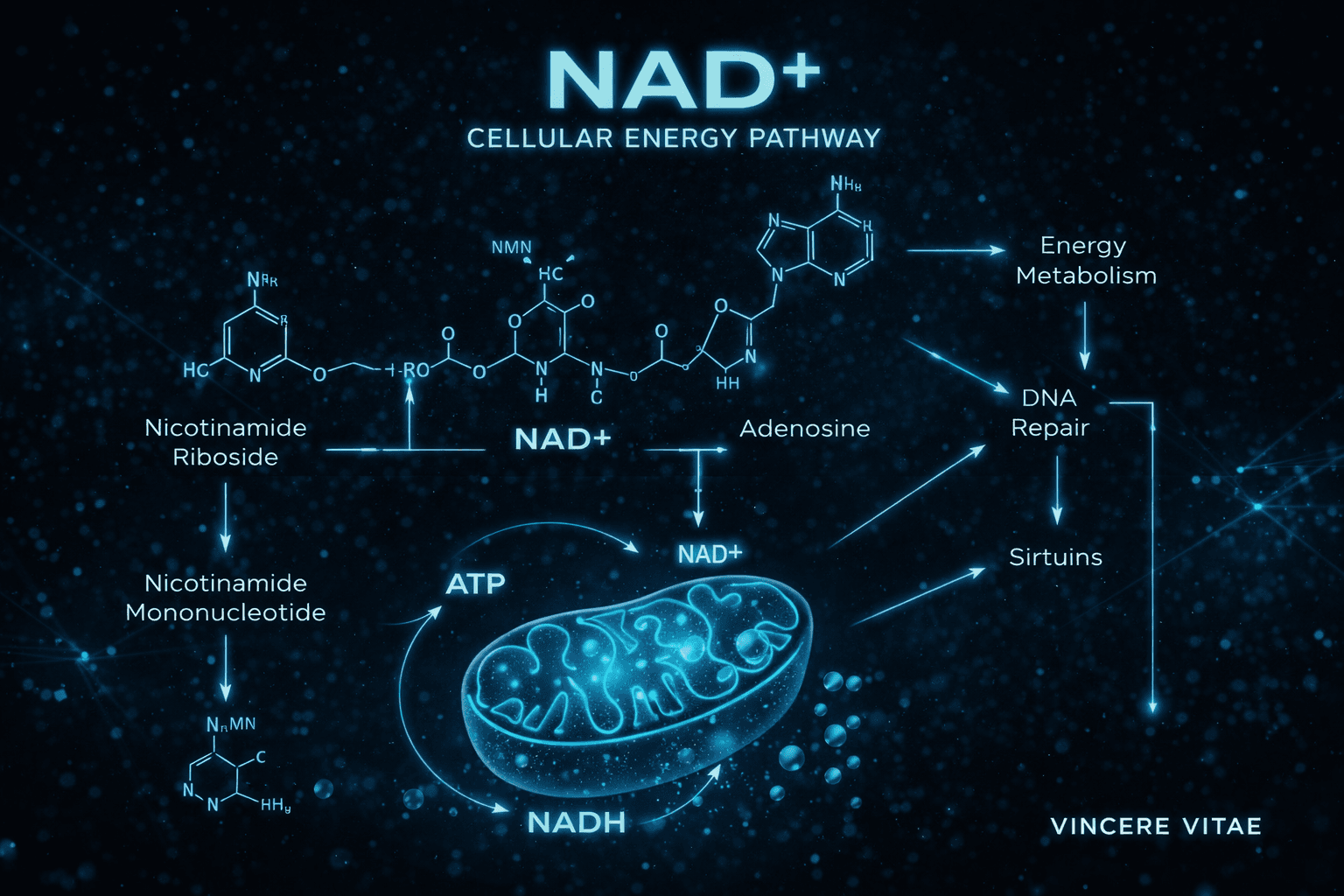
NAD+ in Cellular Energy Metabolism
One of the defining features of NAD+ biology is its role in energy conversion. NAD+ accepts electrons during glycolysis and the tricarboxylic acid cycle, becoming NADH. NADH then donates those electrons to the mitochondrial electron transport chain, helping drive ATP synthesis through oxidative phosphorylation.
This pattern makes NAD+ indispensable for mitochondrial respiration. When NAD+ availability is reduced, cells may experience impaired redox balance, lower metabolic efficiency, and reduced capacity to respond to energetic demand. For this reason, NAD+ is often studied as a central determinant of cellular bioenergetic performance.
In practical research terms, NAD+ helps explain how cells convert nutrients into usable energy while coordinating those processes with broader adaptive signaling networks.
Mechanism of Action
NAD+ functions through two main biological mechanisms. The first is electron transfer in redox metabolism, where it cycles between NAD+ and NADH to support oxidative energy production. The second is substrate-dependent signaling, where NAD+ is consumed by enzymes such as sirtuins and poly(ADP-ribose) polymerases during cellular regulation and repair.
This dual mechanism is one reason NAD+ is so important in research. It is not simply a metabolic cofactor, but a molecule that links energy status to transcriptional control, genomic maintenance, mitochondrial quality, and stress resilience.
Because these systems are tightly interconnected, NAD+ has become a major framework molecule for studying how metabolism and cellular repair are coordinated under both normal and pathological conditions.
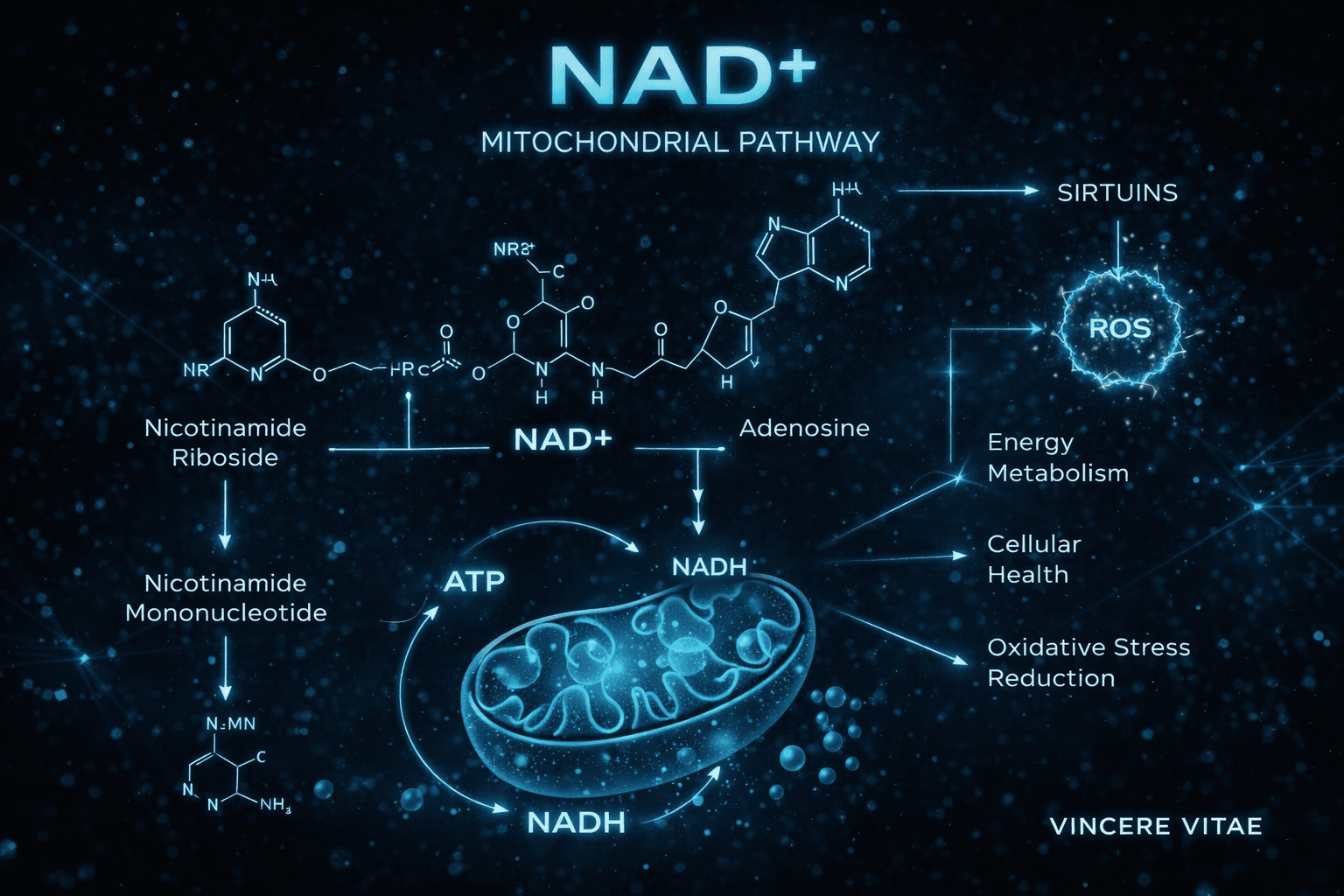
Sirtuin Signaling
A central theme in NAD+ research is its relationship with sirtuins, a family of NAD+-dependent enzymes involved in mitochondrial biogenesis, stress resistance, inflammatory regulation, and metabolic adaptation. Because sirtuins require NAD+ as a substrate, their activity is closely tied to cellular NAD+ availability.
This has made NAD+ especially important in research models focused on longevity biology and mitochondrial resilience. Many investigations explore how NAD+-dependent sirtuin activation may influence energy efficiency, oxidative stress handling, and the preservation of cellular function during aging.
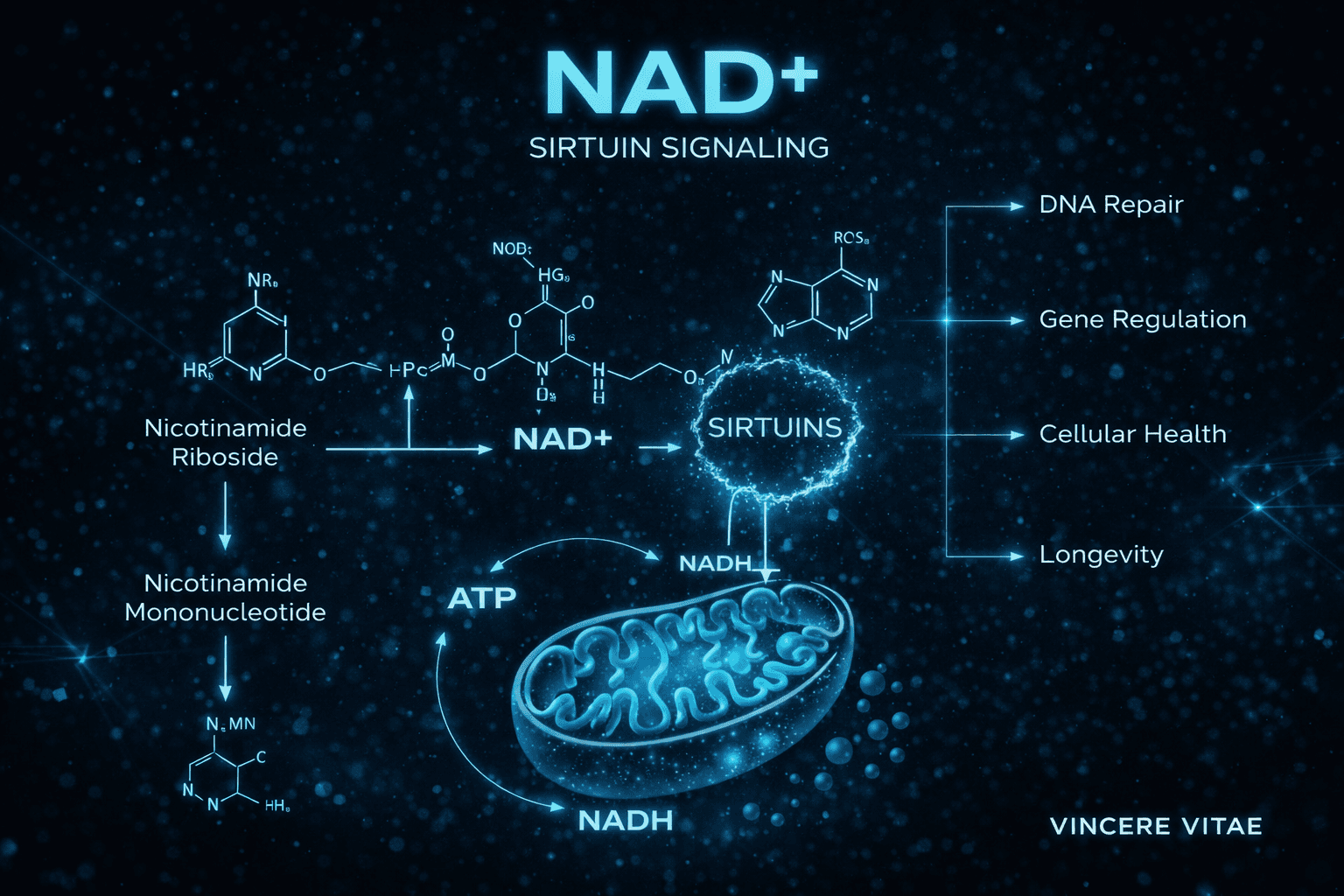
DNA Repair and Genomic Stability
NAD+ is also required for the activity of poly(ADP-ribose) polymerases, commonly referred to as PARPs. These enzymes are part of the cellular DNA damage response system and consume NAD+ to help coordinate repair signaling after genomic insult.
This role makes NAD+ highly relevant in research involving genomic maintenance, oxidative stress, and age-related cellular decline. Under conditions of chronic stress or repeated DNA damage, elevated PARP activity may contribute to NAD+ depletion, which can potentially shift the cell toward lower energetic efficiency and greater metabolic strain.
Age-Related NAD+ Decline and Metabolic Stress
One reason NAD+ has attracted so much scientific attention is that intracellular levels appear to decline with age and chronic metabolic stress. This decline is thought to reflect a combination of increased NAD+ consumption, inflammatory signaling, DNA repair demand, and reduced biosynthetic efficiency.
As NAD+ pools decrease, tissues with high energetic demand may be especially affected. Researchers frequently discuss this in the context of mitochondrial dysfunction, impaired recovery, and reduced cellular resilience across aging models.
NAD+ Compared With Other Research Frameworks
NAD+ is often discussed alongside other molecules involved in mitochondrial biology, longevity science, and cellular metabolism. Comparing these frameworks helps clarify why NAD+ remains one of the most foundational subjects in metabolic research.
Current Clinical and Experimental Research
NAD+ has become a major topic in both experimental and translational research. Investigations span mitochondrial function, age-related decline, exercise adaptation, neurobiology, metabolic disease, and cellular repair systems. Because NAD+ influences such a broad range of biological processes, it is often treated as a central node rather than a narrow pathway-specific factor.
Its importance within current research lies in the fact that it provides a unifying framework for understanding how redox balance, bioenergetics, and adaptive signaling interact. This broad relevance has kept NAD+ central to ongoing scientific interest across multiple fields.
As research continues, NAD+ remains one of the most important biochemical systems for studying the intersection of metabolism, aging, and cellular resilience.
Current Research Limitations
Although interest in NAD+ is substantial, several important research questions remain unresolved. These include the precise contribution of different NAD+-consuming enzymes to age-related decline, tissue-specific responses to changing NAD+ availability, and how long-term modulation of NAD+ systems affects complex physiology over time.
These uncertainties are part of what makes NAD+ such an important scientific topic. It is relevant not only because of its central metabolic function, but also because it may reveal broader principles about how energy and repair systems interact across the lifespan.
Related Research Topics and Internal Reading
For broader context, researchers often compare NAD+ with other compounds and pathways involved in mitochondrial function, peptide signaling, and metabolic adaptation. Related materials across the Vincere Vitae research library can help place NAD+ within a wider investigational framework.
Product
NAD+ Research Product
View the research material listing associated with this review.
Related Research
MOTS-c Research Overview
Explore another mitochondrial-focused compound discussed in metabolic resilience research.
Related Research
Epitalon Research Overview
Review research focused on cellular aging, longevity biology, and related signaling pathways.
Related Research
Semax Research Overview
Compare with a separate research category involving neural signaling and neurobiological investigation.
Research Material Presentation
For research catalog presentation, a clean NAD+ vial image can help visually distinguish the compound while maintaining a scientific layout consistent with the rest of the page.

Explore NAD+ Research Material
Vincere Vitae provides research-focused NAD+ material presented in a format designed for laboratory and educational review. Each listing is structured to support scientific investigation of mitochondrial function, redox balance, and longevity-associated cellular pathways.
View NAD+ Research Product →Frequently Asked Questions
What is NAD+?
NAD+ is a core biological coenzyme involved in redox reactions, mitochondrial ATP production, cellular signaling, DNA repair, and metabolic regulation.
Why is NAD+ important in research?
NAD+ is important because it sits at the center of cellular energy metabolism and also serves as a substrate for enzymes involved in stress adaptation, genomic maintenance, and longevity-associated signaling pathways.
How is NAD+ connected to mitochondria?
NAD+ functions as an electron carrier in glycolysis and the tricarboxylic acid cycle, helping transfer reducing equivalents into mitochondrial oxidative phosphorylation for ATP generation.
Why do researchers discuss NAD+ in aging biology?
Researchers study NAD+ in aging because intracellular levels appear to decline over time, and this decline may affect mitochondrial efficiency, DNA repair capacity, cellular resilience, and metabolic homeostasis.
Selected Research References
- Verdin E. NAD+ in aging, metabolism, and neurodegeneration. Science.
- Yoshino J, Baur JA, Imai SI. NAD+ intermediates: the biology and therapeutic potential of NAD+ metabolism. Cell Metabolism.
- Covarrubias AJ, Perrone R, Grozio A, Verdin E. NAD+ metabolism and its roles in cellular processes during ageing. Nature Reviews Molecular Cell Biology.
Conclusion
NAD+ remains one of the most important molecules in cellular metabolism and signaling research. Its role in redox biology, mitochondrial energy production, DNA repair, and sirtuin-dependent signaling makes it highly relevant to research involving aging, resilience, and metabolic performance.
As scientific investigation continues, NAD+ is likely to remain central to the study of how cellular energy systems, stress responses, and repair pathways interact. Its importance lies not only in what it does directly, but in what it reveals about the deeper architecture of human cell biology.
Research Use Only: This material is provided for laboratory research purposes only. NAD+ material is not approved for human consumption or therapeutic use. All materials are intended for use by qualified professionals in controlled research settings.